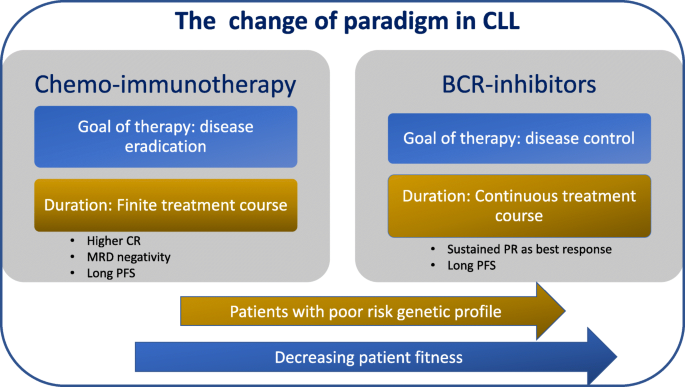
Were busy women and we dont have the time to tend to our hair every single day says Kathy Combs director. Shop Creator Collective.
Unlike other keratin treatments Lasio is safe to use the same day as color services.

Cezanne keratin treatment. Cezanne Instant Keratin Smoothing Treatment is an ultra-fast spray-in smoothing treatment that brings hair to its ideal most beautiful condition in an instant without damaging the hair or using dangerous chemicals. The application process is a little bit longer because you have to leave the product in the hair for 30. Mommy loves you I searched for something that could be applied sans protective face gear.
Cezanne first burst onto the scene in 2014 as a formaldehyde-free option for women who want to wear their locks straight curly or in-between without having to worry about frizz. Glenn Ellis breaks down the three types of keratin treatments and which keratin treatment is best for your hair type. We also recommend the treatment.
But the treatment delivers serious shine and a dramatic reduction in frizz. Cezanne Express Keratin Smoothing Treatment is applied to damp hair processes for 15 minutes and follows the rinse blow dry and flat iron steps as the rest of the treatment formulas. The keratin in the solution gives the hair strength while the Sericin silk proteins lend shine.
The treatment works by bonding liquid keratin to porous areas in the hair shaftthe main culprit of frizz and tanglesleaving hair smoother and silkier than before. Benefits of Cezanne Keratin Hair Treatment Formaldehyde-free Non-drying Customizable depending on desired results Can wash hair immediately post treatment Natural ingredients. It wraps around each hair.
How the treatments work is not through the use of keratin. Cezanne Perfect Finish. And if your client enjoys blow-drying to flat ironing their hair be sure to recommend the Perfect Finish Leave-In to.
Lasio is a heat-activated treatment that infuses the highest quality and most keratin protein into the hair. This treatment option is cost-effective and takes less time than a traditional up to five-month longevity keratin smoothing treatment. Or you could just stick to the Cezanne product line which boasts a complete styling range to ensure proper maintenance after treatment.
This item Cezanne Perfect Finish Keratin Smoothing Treatment 32 Oz 946 ml Cezanne Instant Frizz Eliminator Smoothing Spray 10 FlOz Amazonliss Keratin Smoothing Treatment Hair Straightening - Natural Ingredients - Smooths Strengthens Softens Moisturizers Adds Shine Reduces Frizz 845 fl. The amount of time results lasts will vary by hair type texture and maintenance routine. The Cezanne is the most natural and formaldehyde conscious of the bunch.
Use this anti-aging intensive treatment 1-2 times per week as needed to drench and nourish the hair leaving it silky and youthfully restored. Enter the Cezanne from the Nunzio Saviano salon. Keratin treatments are a semi-permanent hair straightening treatment that smoothes and adds shine to frizzy hair says Fitzsimons.
Sericin produced by a silkworm. A Cezanne Keratin Smoothing treatment will keep your hair luxuriously silky smooth for up to three to five months. As with any smoothing treatment the longevity will vary depending on hair type and maintenance.
As used exclusively with Cezanne Perfect Finish Keratin Smoothing Treatment s unique and proprietary blend of ingredients sericin is able to protect the bonds of the hair in their new form locking the hair into its new smoother straighter shape. The Cezanne treatment is 350-450 can last for months and takes up to two and a half hours as compared to 200 for Julien Farels which lasts for up to two months and takes 45 minutes. The Cezanne treatment is formaldehyde-free and employs a different active ingredient.
When I inquired about the formaldehyde-free keratin treatment advertised on its website I learned it was a formula called Cezanne that promises to smooth and de-frizz hair while restoring shine cut blow-drying time in half and most importantly work on all hair types. This popular conditioning treatment makes kinky frizzy or difficult curly hair soft manageable and beautifuland can reduce your daily. It claims to do everything a keratin treatment.
Results last up to 4 weeks. Keywords keratin hair treatment More from Allure. Reboot is a replenishing and restorative treatment to extend the life of your Cezanne keratin smoothing treatment.
We can provide you with a gorgeously rejuvenating take-home care pack designed to lock in the effects for as long as possible. To protect the treatment be sure to send your client home with Cezannes Perfect Moisture Shampoo and Conditioner for proper maintenance.